|
Home Page
Known About Fluoride
Toothpaste Warning
Interferes with Brain
Fetal Vulnerability
Neurotoxicity
Needs to Be Known
Fetal Fluoride Intake
Fluoride Drugs
Amniotic Fluid
Bogus Sales Pitch
Oregon Deception
Crazy Advice
Premature Birth
Infants Overdosed
Infant Mortality
Life Decay
Mental Retardation
Cancer
Mental Fluorosis
Fluoride and
Neurodegeneration
End Fluoridation
Save $Billions
Know News
Prenatal Microbiome
Fluoride & Obesity
Vascular Dementia
Arterial Stiffness
2019 Studies
Fluoridation 'Science'
CDC's Irrationale
EPA's Pollutant
PHS's Debunkery
The Book
Contents
Author
|
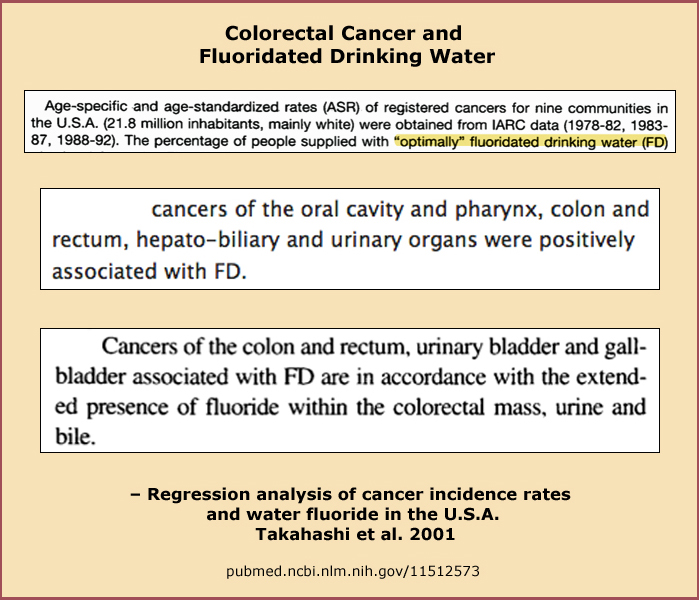
Fluoridated Water and Cancer
Death rates in the US from cancer (malignant neoplasms, the #2 leading cause of death in 2013) average 7.6% higher in the 10 most fluoridated states compared to the 10 least fluoridated states.
Reduced Adhesion Forces are Involved in Cancer Metastasis
"Tumor cells that exhibit low cell-cell adhesion forces would represent a highly metastatic phenotype since they detach more easily." [Studying Adhesion Forces Between Cancer Cells 2014]
In the majority of the human cancers, it has been clearly demonstrated that alterations in adhesion forces are implicated in the invasion and metastasis process. Metastasis, the late stage of cancer spread, involves the migration of malignant neoplasms from one anatomical site to another. This process is likely to be most affected by de-arrangement or alterations in adhesion receptor levels or function. [Golias et al. 2005]
|
Fluoride Reduces Adhesion Forces
Researchers in Experimental Physics at Saarland University* in Germany recently discovered how fluoride disrupts cavity-causing bacteria. Fluoride reduces the adhesion forces of bacteria, so they are less able to adhere to teeth, where they produce the acid that causes cavities.
Because fluoride reduces the ability of decay-causing bacteria to stick to teeth, it is easier to wash away these bacteria by saliva, brushing, and other activity. [Loskill et al. 2013] The researchers said, "Fluoride appears to weaken bacterial adhesion forces in general."
|
Breast cancer metastasis is linked to Sevoflurane anesthesia. [Li et al. Jan. 2020]
Sevoflurane is a fluorinated general anesthetic that significantly
increases blood fluoride levels. [Goldberg et al. 1996]
Sevoflurane is also associated with post-operative delirium and in
endothelial dysfunction which plays an early pathogenic role in many diseases.
Fluoride Decreases Expression of Neural Cell Adhesion Molecules
Neural cell adhesion molecules are widely expressed in the nervous system, where they are involved in axon growth and guidance – fundamental processes that underlie formation of the synaptic connections and myelinated nerve structure crucial to brain development.
Fluoride exposure has been shown to cause a dose-dependent decrease in expression levels of neural cell adhesion molecules (NCAM) in rat hippocampal neurons. In particular, the NCAM-140 protein expression level was significantly lower in response to the lowest dose of fluoride used. [Xia et al. 2007; Zhang et al. 2007]
NCAM-140 is found in migrating growth cones that are crucial to the formation of synaptic connections. [Beggs et al. 1997]
Autism Spectrum Disorder (ASD)
Research suggests that brain enlargement in autism is likely due to cell adhesion dysfunction. [Lahiri et al. 2013] Significantly lower serum levels of several types of adhesion molecules, including NCAM, have been found in ASD. [Kameno et al. 2013; Plioplys et al. 1990]
Neural pathways involving synaptic cell adhesion are disrupted in some people with autism, including alterations in the structure and expression of NCAM. [Betancur et al. 2009; Sakurai 2012]
– Pregnancy and Fluoride Do Not Mix, page 52
|
*In 2010, Saarland University researchers showed that the long-hypothesized "protective layer" that fluoride supposedly forms on teeth is in fact only six nanometers thick. (10,000 layers would span the width of a human hair.) Such a minuscule layer is quickly worn away by ordinary chewing, so would not shield teeth from decay. – Does fluoride really fight cavities by 'the skin of the teeth?' [American Chemical Society]
This confirms what the CDC already knew: "The prevalence of dental caries in a population is not inversely related to the concentration of fluoride in enamel, and a higher concentration of enamel fluoride is not necessarily more efficacious in preventing dental caries."
What's more, there is no evidence of any better oral health in the eight million Americans the CDC said are served by community water systems that have "sufficient naturally occurring fluoride concentrations." [CDC 2008]
|